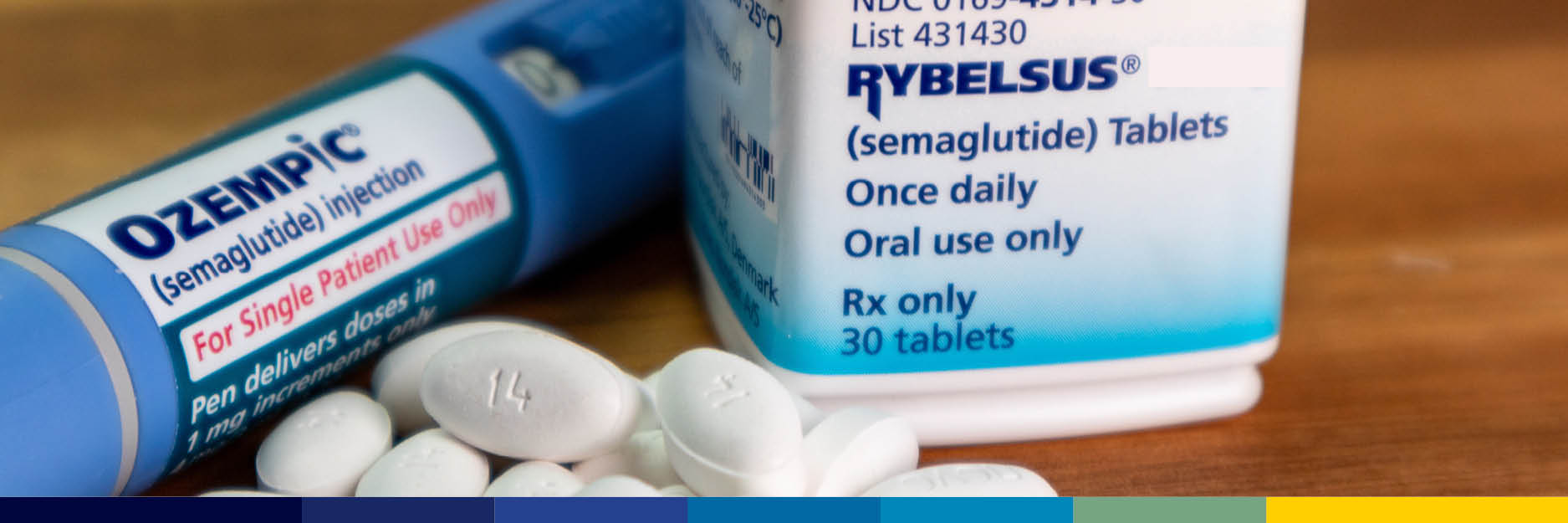
Novo Nordisk recently received FDA approval to bring its oral semaglutide formulation, Rybelsus, under the Ozempic brand – aligning oral and injectable semaglutide for type 2 diabetes under a single, highly recognized name.
This isn’t a new molecule – it’s a strategic rebrand aimed at:
• Improving patient and provider awareness
• Reducing brand fragmentation across GLP‑1 therapies
• Simplifying prescribing, education, and prior authorization workflows
As GLP‑1 utilization continues to grow, branding decisions like this have real implications for access, utilization management, and patient understanding.